For example, if you have 1.5 mol of NaCl dissolved in 0. It is important to remember that mol in this expression refers to moles of solute and that L refers to liters of solution. The accompanying table lists some useful hydrates. Sodium chloride, also known as salt, common salt, table salt or halite, is an ionic compound with the chemical formula NaCl, representing a 1:1 ratio of. Molar mass can then be used as a conversion factor to convert amounts in moles to amounts in grams. Use the periodic table to find the molar mass of hydrogen, H. :max_bytes(150000):strip_icc()/PeriodicTable-56a12c983df78cf772682271.png)
Aluminum chloride hexahydrate is an active ingredient in antiperspirants. What is the molar mass of salt, NaCl Na: 22.99g/mol. This relation provides a conversion factor to go from grams to moles. Thus, one mole of CO 2 weighs 44.01 grams. Epsom salt (MgSO 4♷H 2O) is used as a bathing salt and a laxative. The formula mass of CO 2 is: 12.01 + 2 (16.00) 44.01. Sodium chloride /sodim klrad/, commonly known as table salt, is an ionic compound with the chemical formula NaCl, representing a 1:1 ratio of sodium. ½H 2O), known as plaster of Paris, is used to make casts for broken bones.Hydrates have various uses in the health industry. A compound is 42 sodium, 19 phosphorus, and 39 oxygen by mass. 
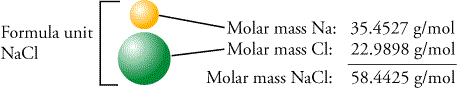
What is the percent by mass of chlorine (Cl) in sodium chloride 35.5 60.7 0.606. What is the percent by mass of sodium (Na) in sodium chloride 39.3 22.9 0.393. The name of this compound is copper(II) sulfate pentahydrate, with the penta- prefix indicating the presence of five water units per formula unit of copper(II) sulfate. Use the periodic table to find the molar mass of NaCl. The hydrate of copper(II) sulfate has five water units associated with each formula unit, so it is written as CuSO 4♵H 2O. For example, on the pre-1961 atomic weight scale the molecular weight of table salt, Sodium chloride NaCl would have been 58.45. The two chemical formulas are separated by a vertically centered dot. To write the chemical formula of a hydrate, write the number of water units per formula unit of compound after its chemical formula. The molar mass of an element is the same value as its atomic weight listed on the periodic table except it has the units g/mol. Multiply the atomic weight (from the periodic table) of each. Hydrates are solids, not liquids or solutions, despite the water they contain. Use the chemical formula to determine the number of each type of atom present in the compound. These compounds, called hydrates, have a characteristic number of water units associated with each formula unit of the compound. Some ionic compounds have water (H 2O) incorporated within their formula unit.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
